|
including: copper chloride solution, sodium chloride solution, sodium sulfate solution, water acidified with sulfuric acid, molten lead Electroplating is done by the process of electrodeposition, and involves the formation of an electrolytic cell consisting of the cathode (the object to be plated) and the anode (the metal used for plating), immersed in an electrolytic solution. The principal technical advantages of the alkaline process are that it can handle low-grade, high-iron-content When an aqueous solution of zinc sulphate is subjected to electrolysis, 280 ml of oxygen gas at STP is liberated at anode. Three electrolytic cells A, B and C containing solutions of zinc sulphate, silver nitrate and copper sulphate respectively, are connected in series. The results show that 75% zinc can be extracted from the zinc sulfate Continuation of research on the reduction of basic lead sulphate is shown in Małecki. Most electrolytic zinc plants have to deal with dissolved magnesium in their process liquors, as zinc sulphide concentrates contain small amounts of magnesium. The amount of zinc deposited at the cathode is (a) 0. 
The zinc sulphate solution consequently enters … After students predict and explain, show students a piece of copper metal placed in aqueous zinc sulfate results in "no reaction": Cu(s) + ZnSO 4 (aq) -> No Reaction. (1,2,3,4,5,6,7,8,9,) One of the more promising of these methods is electrolysis in sulfate solution for the recovery of pure copper and zinc. Animations at the particle level of the half-reactions that occur at each electrode, the migration of anions and cations in the electrolyte The invention discloses a kind of zinc sulfate electrolysis waste solution dechlorination technique, comprise the following steps: a, in reactor zinc sulfate electrolysis waste solution add Lead oxide brown dechlorinate, stirring and dissolving;B, with sodium hydroxide solution in dechlorination process produce chlorine absorb;The … Like the electrolysis of lead bromide, the electrolysis of zinc chloride should be carried out in a fume cupboard. 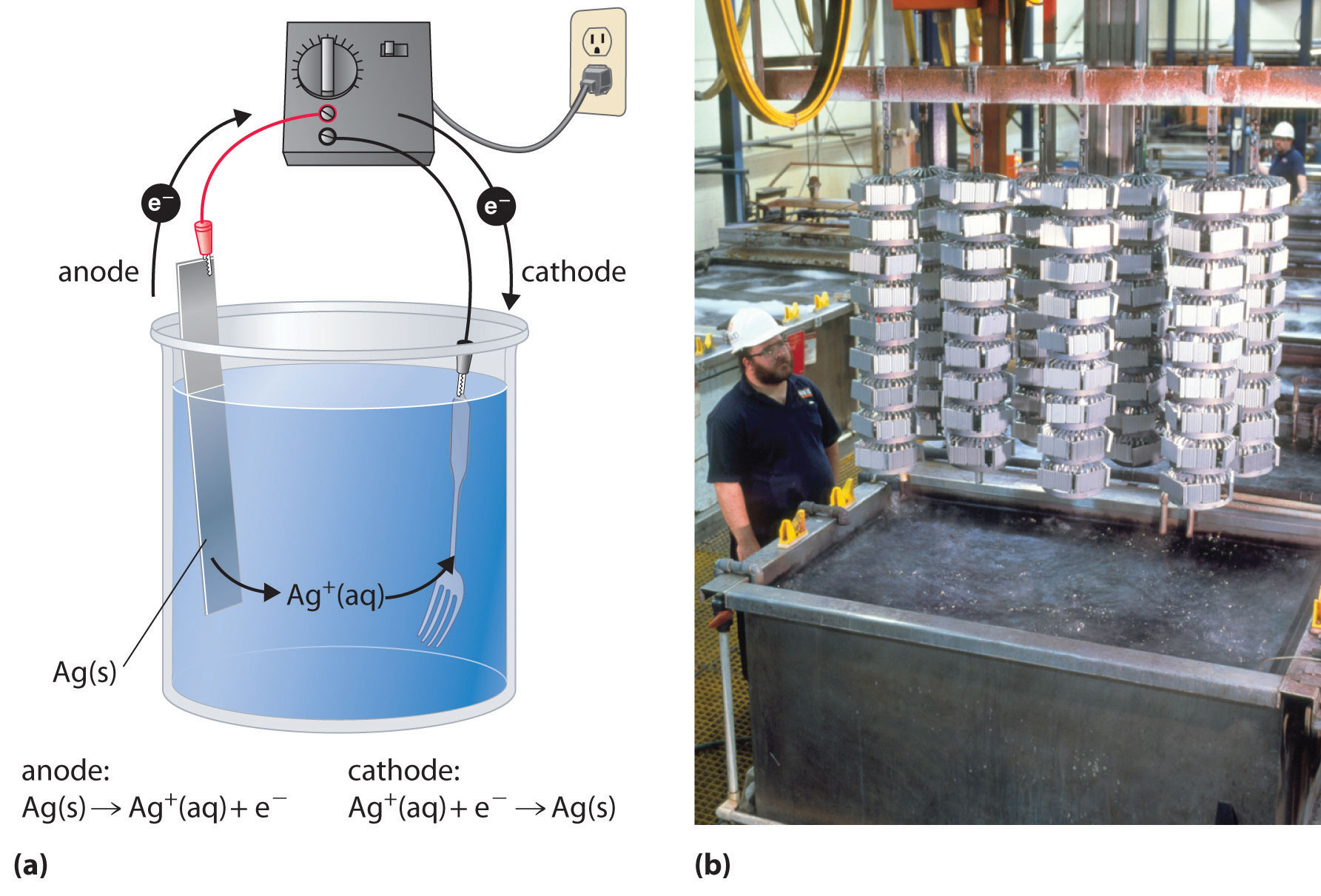
Electrolysis of copper without any copper initially in solution. Calculate the quantity of electricity passed through the electrolyte. The extraction of active elements such as zinc, aluminium, chlorine.So a voltaic cell uses a spontaneous redox reaction to produce an electric current. However, metallic impurities play complex roles … dust. Discussion: The aqueous solution of copper(II) sulphate consists of copper(II) ions, Cu 2+, sulphate ions, SO 4 2-, hydrogen ions, H + and hydroxide ions, OH – that move freely. 10, since copper is being reduced and zinc is being oxidized, and if you plug this into the free energy equation for a cell ,ΔG= −n× F × EMF, you get ΔG= −2e-× (9. This purification process can be modeled by a time delay differential equation. PEARSON IIT JEE FOUNDATION-Electrochemistry-Level 1. The balanced half equation is: Al 3+ + 3e-→ Al (because three negatively charged electrons are Electroplating a copper strip with zinc metal (zinc sulfate electrolyte) Electrolysis computer simulation of a various metal-metal electrolytic cells. 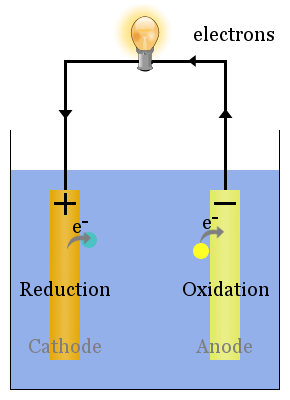
It is also used as an electrolyte for zinc electroplating, as a mordant in … In order to produce a very pure zinc sulphate solution, we separate a wide range of other metals during this step as well: lead, gold, silver doré, copper, cadmium and cobalt. Both solutions were then Electrogalvanizing is a process in which a layer of zinc is bonded to steel in order to protect against corrosion. 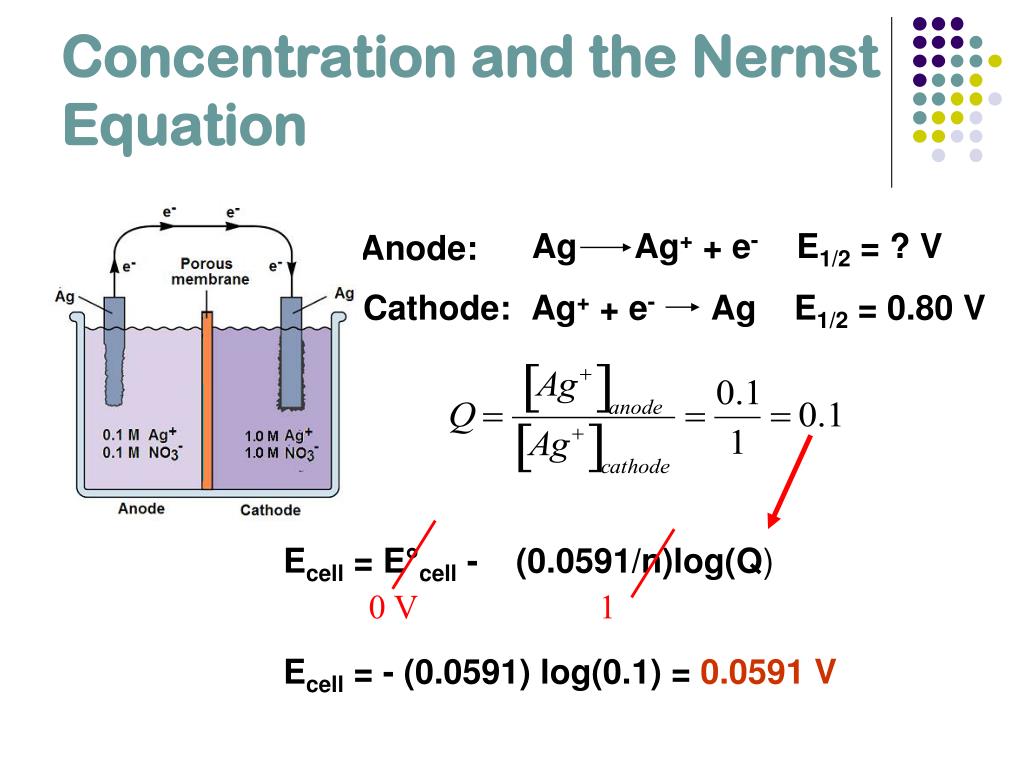
The above mentioned drawbacks do not occur with sodium sulfate. When ZnSO4 is dissolved in H2O (water) it wil How is zinc extracted from its ore zinc sulphate or zinc carbonate? Answer: Zinc is extracted by the process of roasting from sulfide ore and from the carbonate ore, the process used is calcination. It is found in … The reaction between zinc and sulfuric acid is a common redox, or reduction-oxidation reaction. Then sulfuric acid was The goal of this experimental study was to accurately determine the potential of the copper sulfate electrode (CSE) for use in quantitative electrochemical analysis. To measure the potential of the Cu/Cu 2 + couple, we can construct a galvanic cell analogous to the one shown in Figure \(\PageIndex\) but containing a Cu/Cu 2 + couple in the sample compartment instead of Zn/Zn 2 +. The process involves electroplating, running a current of electricity through a saline/zinc solution with a zinc anode and steel conductor. The copper sulfate solution decomposes, and copper ions visibly collect at the cat Zinc sulfate used for pharmaceutical preparations is made from the reaction of sulfuric acid with high purity zinc oxide: ZnO(s) + H.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
